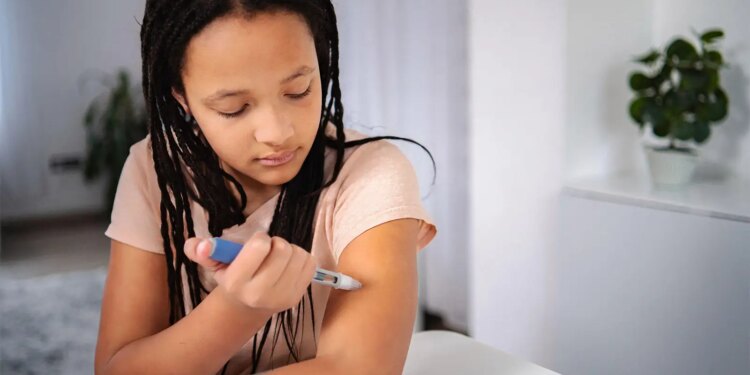
Two insulin-dependent adolescents with type 1 diabetes and obesity had metabolic improvements after starting a low-dose GLP-1 agent, a case series showed.
One year after adding injectable semaglutide (Ozempic, Wegovy) to lifestyle intervention, a 17-year-old female with a 10-year history of type 1 diabetes lost 12 kg (26.5 lb) and reduced her body mass index (BMI) from 30.1 to 26, according to Julie Kloppenborg, MD, PhD, of Steno Diabetes Center Copenhagen in Denmark, and colleagues.
Her HbA1c dropped by 2.5%, her total daily insulin dose decreased by 27.7 units, and her time spent in target range modestly increased, they wrote in Pediatrics.
The second patient, a 12-year-old female with a 4-year history of type 1 diabetes, had similar benefits. She decreased her body weight by 8.4 kg (18.5 lb), her BMI from 38 to 34.1, her total daily insulin dose by 53 units, and had a modest HbA1c improvement. She also increased her time-in-range by 9 percentage points, and decreased her time spent below range (<70 mg/dL) and above range (>180 mg/dL) by 4 percentage points each.
Neither patient experienced severe hypoglycemia (<54 mg/dL) and no significant adverse events occurred.
“These two cases illustrate the growing burden of obesity in adolescent out-patient clinics handling type 1 diabetes and the urgent need for evidence-based targeted interventions addressing both weight management and glycemic control in this challenging population,” Kloppenborg and co-authors highlighted.
While diabetes technology like continuous glucose monitoring (CGM) has transformed care, Kloppenborg’s group said that technology alone often fails to optimize HbA1c when obesity is present. “As obesity further complicates type 1 diabetes by exacerbating the inherent cardiovascular risk, novel interventions are required to improve outcomes in this growing population,” they wrote.
Christopher Romero, MD, of the Icahn School of Medicine at Mount Sinai in New York City, agreed that conventional treatments are often inadequate for patients with type 1 diabetes and obesity. “It is important to consider other interventions for our type 1 diabetes population as a means to ease their medication burden of insulin, and more importantly optimize their health,” he told MedPage Today.
Romero, who wasn’t involved with the study, warned that poorly controlled diabetes in youth carries a higher risk for both macro- and microvascular complications in early adulthood.
GLP-1 agents are widely used therapies for type 2 diabetes and obesity, including in adolescents, but their use in type 1 diabetes has been limited due to safety concerns. Labels currently warn of increased risks for hypoglycemia in patients on insulin, and no GLP-1 agents are currently indicated for type 1 diabetes.
“Hypoglycemia is not typically seen in our patients with type 2 diabetes, but given that the vital therapy for type 1 patients is insulin continuously, there is already an inherent risk for hypoglycemia,” noted Romero.
In these two cases, adding GLP-1 therapy was considered only after years of “limited success” with lifestyle intervention and increasing insulin dosages, the researchers said. Risk mitigation for both patients included prandial insulin down-titration, reinforced CGM alerts, ketone monitoring, and structured hypoglycemia education before initiating semaglutide.
Doses were titrated based on clinical response and tolerability, and escalations were postponed if side effects occurred. Both patients reached a 0.5 mg dose by month 6.
Prior data on GLP-1 drug use in type 1 diabetes is limited. A small trial found semaglutide (1 mg) reduced glucose levels and weight in patients with type 1 diabetes and obesity without safety concerns. A small real-world study of adolescents and young adults with type 1 diabetes also reported metabolic improvements after 10 months, though safety wasn’t addressed.
Despite limited data, a nationwide study found an uptick in GLP-1 agent prescribing for type 1 diabetes patients in recent years.
Larger clinical trials are urgently needed, Kloppenborg and colleagues said. “Confirming the efficacy and safety … could significantly enhance treatment options for adolescents with type 1 diabetes and obesity, ultimately improving their long-term metabolic and cardiovascular health trajectories,” they wrote.
Romero agreed, noting that because insulin requirements vary widely in children based on age, size, and pubertal status, “this potentially makes dosing of a GLP-1 agonist more complicated and requires well-thought-out research studies.”
Both patients remain under gradual dose up-titration in close collaboration with pediatric diabetologists and clinicians experienced in GLP-1 receptor agonists, the researchers noted.
Source link : https://www.medpagetoday.com/endocrinology/type1diabetes/121164
Author :
Publish date : 2026-05-08 04:01:00
Copyright for syndicated content belongs to the linked Source.