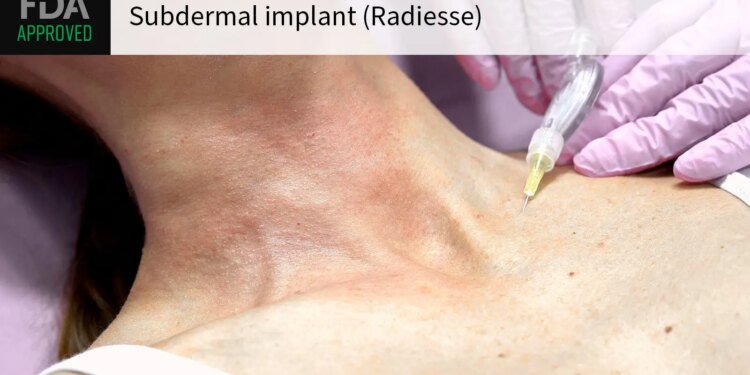
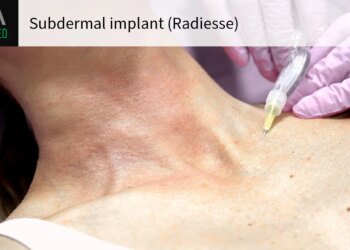
The FDA approved a subdermal implant (Radiesse) for filling wrinkles and providing volume in the so-called décolleté area — the area around the neck and upper breasts — despite concerns that materials in the product could interfere with breast imaging.
The dermal filler, approved for adults 22 and up, contains hydroxylapatite microspheres that can be visible on x-rays and CT scans and potentially obscure imaging, a concern that warranted an FDA advisory committee meeting on the product last year.
“As a breast imager, my focus is to find a cancer as small as possible,” panelist Sandra Shuffett, MD, of Baptist Health Medical Group in Lexington, Kentucky, said at the meeting. “That is my concern, with the fillers potentially obscuring a cancer on a mammogram until it grows larger and then requires more serious treatment.”
In its approval announcement, Merz Aesthetics said a study showed its product was not visible on mammograms or breast ultrasounds after treatment in the décolleté area. But the company said it’s “very important” that patients inform their doctors if they received the dermal filler in the décolleté area, and that patients should tell their radiologist in order to ensure appropriate image interpretation.
FDA is requiring a 30-patient postmarketing study to determine if treatment in the décolleté area interferes with radiographic imaging of breast tissue. Women will receive three dermal filler treatments, 6 weeks apart, with imaging performed at baseline and 1 month after the third injection.
Merz Aesthetics touts its dermal filler as a “regenerative biostimulator” that works by stimulating the production of collagen, elastin, and other proteins to help firm and tighten skin.
According to the company, pivotal trial data supporting the new approval showed that after a year from initial treatment, more than 80% of patients felt satisfied with skin tightness and 83% said they would undergo treatment again. Additionally, 90% of healthcare providers reported visible improvements by 4 months.
The product is also approved for use in treating wrinkles of the hands or face.
Common adverse events in trials of the hands or face included bruising, redness and swelling, pain, itching, nodules at the injection site, loss of sensation, difficulty chewing or performing other activities, and other local side effects. Bruising or bleeding may be more common in patients on blood thinners, and delayed-onset inflammation can occur.
The company also warned that patients with a history of herpes could have a breakout and said that patients should minimize strenuous activity and avoid extensive sun/heat the day after treatment and until any swelling or redness resolves.
Source link : https://www.medpagetoday.com/dermatology/generaldermatology/120699
Author :
Publish date : 2026-04-08 16:40:00
Copyright for syndicated content belongs to the linked Source.