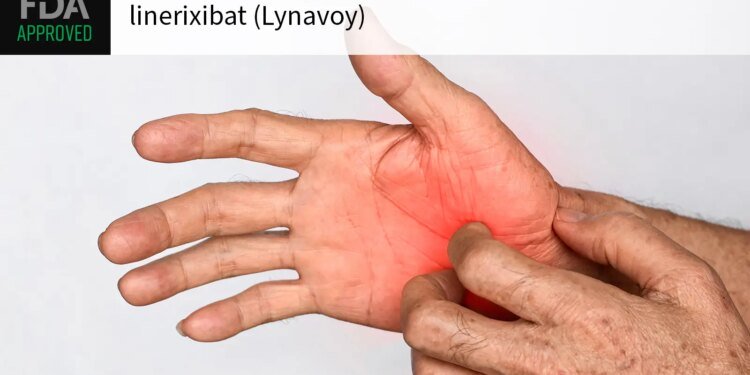
The FDA approved linerixibat (Lynavoy) as the first drug indicated for cholestatic pruritus stemming from primary biliary cholangitis (PBC), drugmaker GSK announced on Thursday.
PBC is a rare but serious autoimmune disease where bile flow from the liver is disrupted. The vast majority (89%) of PBC patients experience an internal itch — thought to result from bile acids in circulation — that affects sleep, quality of life, and is sometimes so debilitating it can necessitate transplant even in the absence of liver failure.
An ileal bile acid transporter (IBAT) inhibitor, linerixibat was developed to block bile acid re-uptake and reduce multiple mediators of chronic itch.
“The approval of linerixibat represents an important opportunity to improve the lives of people with PBC … who struggle with uncontrolled and often debilitating pruritus,” said Christopher Bowlus, MD, of the University of California Davis, in a statement. “The impact of itch on people living with PBC can be profound and treatment options have until now been limited. The FDA’s decision marks a major milestone in PBC pruritus care that addresses a critical area of unmet need.”
Approval was supported by findings from the randomized GLISTEN trial. Among 238 patients in the global phase III study, those treated with linerixibat had a significant reduction in itch score from baseline to 24 weeks compared with those treated with placebo, with mean score changes of -2.86 versus -2.15 on a 10-point scale (0 represents no itching and 10 the worst imaginable itching; P=0.001).
The effect was rapid, with a mean change at week 2 of -1.78 with linerixibat versus -1.07 with placebo (P<0.001).
Pruritus-related sleep interference was also reduced with linerixibat (P=0.024), and more patients in the linerixibat group had clinically meaningful itch improvement compared with placebo at week 24 (56% vs 43%, nominal P=0.043).
Common adverse events (AEs) with linerixibat in the study included mostly grade 1/2 cases of diarrhea (61%) and abdominal pain (18%). Treatment discontinuation for each AE occurred in 4%.
As part of the approval, the FDA will require a worldwide postmarketing study for linerixibat’s safety during pregnancy, with infant outcomes followed for at least 1 year, and the agency will evaluate data from an ongoing clinical trial to assess liver injury, bone health, and bleeding risks.
Source link : https://www.medpagetoday.com/gastroenterology/generalhepatology/120396
Author :
Publish date : 2026-03-19 21:23:00
Copyright for syndicated content belongs to the linked Source.