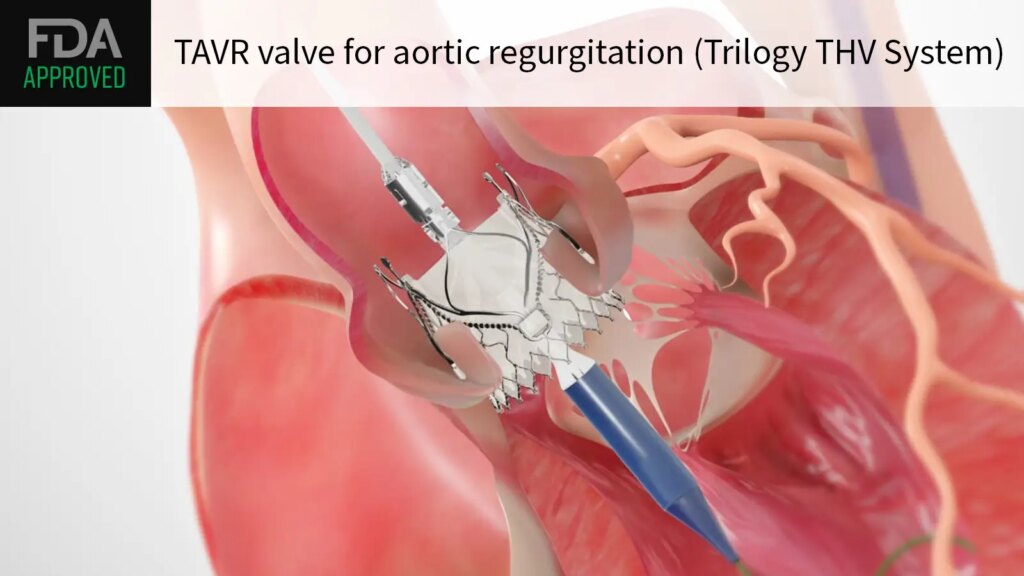
The FDA approved the Trilogy Transcatheter Heart Valve System for the treatment of symptomatic severe aortic regurgitation, device maker JenaValve Technology announced.
This approval makes Trilogy the only transcatheter aortic valve replacement (TAVR) system indicated for patients with symptomatic severe aortic regurgitation who are at high risk for surgical aortic valve replacement.
Unlike other TAVR valves designed for aortic stenosis, the Trilogy system attaches directly to the native aortic leaflets without relying on annular calcification for anchoring. The system is implanted via transfemoral access and features locators that align the device with the patient’s cusps for proper alignment.
“The unique design of the Trilogy System — with its locator technology providing secure anchoring even in the absence of calcium — addresses the fundamental challenge that has made AR [aortic regurgitation] so difficult to treat,” said Martin Leon, MD, of Columbia University Irving Medical Center and NewYork-Presbyterian Hospital in New York City, in the press release. “This approval establishes a new standard of care for high-risk AR patients and is a landmark achievement for the field of interventional cardiology.”
Premarket approval was based on the prospective ALIGN-AR pivotal trial in which the Trilogy system was found to be safe and effective in patients from 20 sites.
The device met the safety benchmark, with a 26.7% rate of combined 30-day all-cause mortality, all stroke, life-threatening or major bleeding, major vascular complications, acute kidney injury or dialysis, valve intervention, new permanent pacemaker, and moderate or worse paravalvular leak (P<0.0001 for noninferiority to the prespecified margin of 40.5%), and also met the primary efficacy endpoint for 1-year all-cause mortality, with a rate of 7.8% that was significantly noninferior compared with the prespecified margin of 25%.
Aortic regurgitation describes a leaky aortic valve that allows backflow from the aorta into the left ventricle during diastole. The condition, also known as aortic insufficiency, has been considered by some to be a “forgotten” heart valve disease due to a general belief of its slow disease progression and common lack of symptoms.
The company announced that the Trilogy system will undergo a limited initial launch at select clinical sites.
The Trilogy is CE Marked in Europe for both aortic regurgitation and aortic stenosis.
Please enable JavaScript to view the comments powered by Disqus.
Source link : https://www.medpagetoday.com/cardiology/interventionalcardiology/120376
Author :
Publish date : 2026-03-18 21:22:00
Copyright for syndicated content belongs to the linked Source.