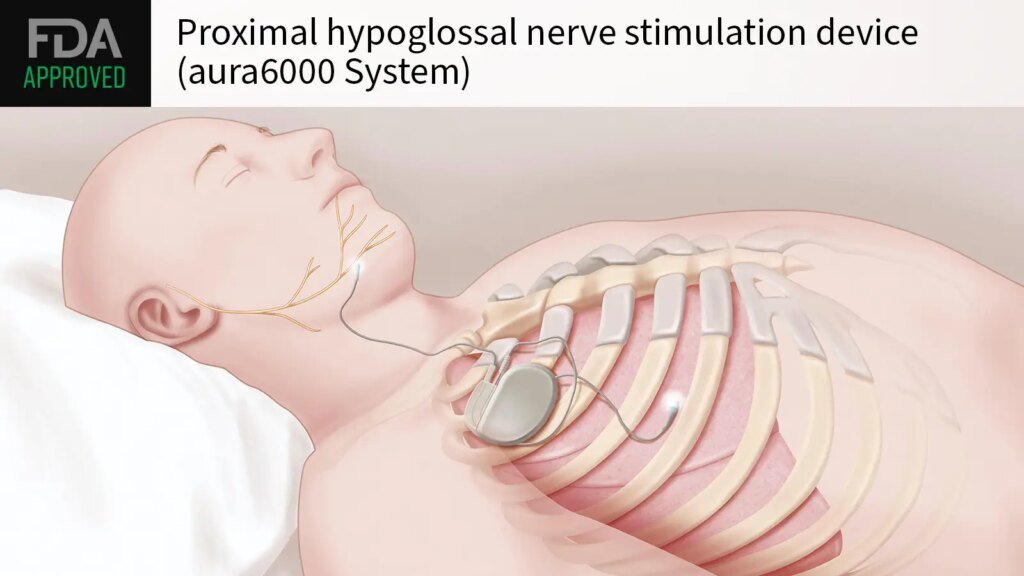
The FDA approved a proximal hypoglossal nerve stimulation device (aura6000 system) for adults with moderate to severe obstructive sleep apnea (OSA), maker LivaNova announced on Thursday.
FDA’s premarket approval — the pathway for class III medical devices — stipulates use in patients with an Apnea-Hypopnea Index (AHI) ranging from 15-65 who have failed or are not suitable for first-line therapies such as a continuous positive airway pressure machine.
In the multicenter randomized OSPREY trial, AHI response rate — a 50% or greater drop, and to below 20 events per hour — was significantly higher in patients receiving active treatment with the proximal hypoglossal nerve stimulator versus a control group, which received an inactive device (58.2% vs 13.5% at 6 months, P<0.001). Patient-reported outcomes for daytime sleepiness and functional outcomes of sleep quality improved with the device at 6 months as well.
At baseline, patients in the active arm had a median 34.3 AHI events per hour, a number that dropped to 11.6 events per hour after 6 months of use and remained stable at 12 months. Topline data showed a 12-month response rate of 65% with the device, along with a 68% reduction in AHI and oxygen desaturation events per hour.
The most common adverse events (AEs) included incision discomfort, stimulation discomfort, and wound healing. No serious AEs occurred attributable to the device.
Drug-induced sleep endoscopy was not required for enrollment in OSPREY, and the trial did not exclude patients with complete concentric collapse. As such, the next-generation device becomes the first hypoglossal nerve stimulation therapy in the U.S. “without complete concentric collapse contraindication or warning language,” according to LivaNova.
The company said it expects to launch the aura6000 system next year.
Please enable JavaScript to view the comments powered by Disqus.
Source link : https://www.medpagetoday.com/pulmonology/sleepdisorders/120422
Author :
Publish date : 2026-03-20 19:48:00
Copyright for syndicated content belongs to the linked Source.