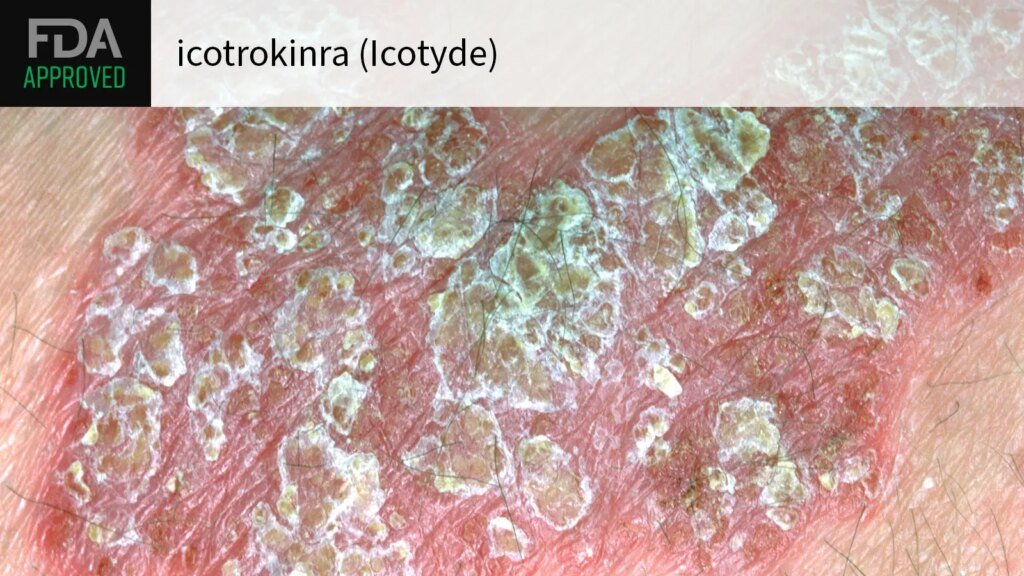
The FDA has approved the first oral interleukin (IL)-23-targeted therapy for plaque psoriasis, Johnson & Johnson announced.
The approval of icotrokinra (Icotyde) stipulates use in adults and children 12 or older with moderate/severe psoriasis who are candidates for systemic therapy or phototherapy.
“Icotyde delivers something unique in psoriasis treatment, combining skin clearance with a favorable safety profile in a once-daily pill,” said Linda Stein Gold, MD, of Henry Ford Health in West Bloomfield, Michigan, in the company statement. “With new guidance from the International Psoriasis Council that clarifies when to move beyond cycling on topical treatments to systemic therapy, an innovative option like Icotyde is a potential game changer for many adult and adolescent patients.”
Leah M. Howard, JD, president and CEO of the National Psoriasis Foundation, added, “Finding the right treatment can take time, during which people with psoriatic disease should be considering multiple factors, from efficacy to safety to how the treatment fits into their everyday life. The approval of a novel systemic therapy changes the conversation about treatment options for our community.”
Evidence supporting the approval came from multiple phase III trials involving 2,500 patients. The recently reported ICONIC-ADVANCE 1 and 2 trials, involving more than 1,400 patients, showed that two-thirds of those treated with icotrokinra had at least 90% skin clearance after 24 weeks versus about 40% for control groups treated with deucravacitinib (Sotyktu). Rates of clear/nearly clear skin by investigator assessment (IGA) showed absolute differences of 15-20% favoring the oral IL-23 receptor antagonist.
The phase III, placebo-controlled ICONIC-LEAD, reported last year at the American Academy of Dermatology meeting, also showed that two-thirds of patients treated with icotrokinra met IGA criteria for clear/nearly clear skin at 4 months.
In clinical trials, the most common side effects associated with icotrokinra were headache, nausea, cough, fungal infections, and fatigue.
Ongoing trials are evaluating the IL-23 inhibitor in psoriatic arthritis, ulcerative colitis, and Crohn’s disease.
Please enable JavaScript to view the comments powered by Disqus.
Source link : https://www.medpagetoday.com/dermatology/psoriasis/120361
Author :
Publish date : 2026-03-18 16:34:00
Copyright for syndicated content belongs to the linked Source.