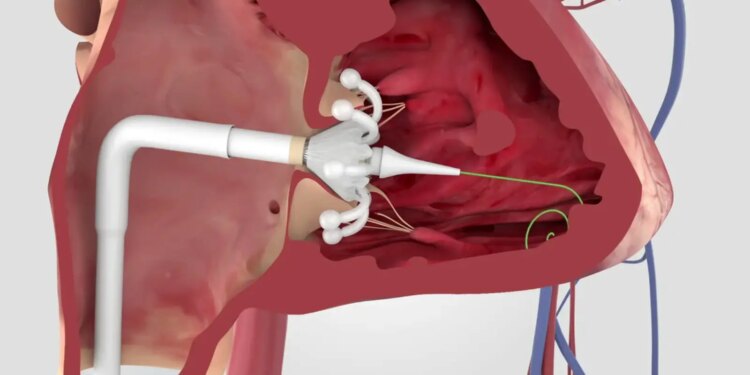
- A nationwide registry captured short-term results of transcatheter tricuspid valve replacement since it debuted in the U.S. in 2024.
- These early results support the device’s safety and effectiveness in clinical practice for patients with severe, symptomatic tricuspid regurgitation.
- Of note, 30-day bleeding was down to 7.9%, and the need for a first cardiac implantable electronic device was 15.9%.
Transcatheter tricuspid valve replacement (TTVR) translated reasonably well from the clinical trial setting to early real-world practice, according to registry data.
In the Society of Thoracic Surgeons/American College of Cardiology (STS/ACC) TVT Registry, event rates at 30 days showed safety and effectiveness outcomes at least consistent with the pivotal trial data for a cohort of older patients with symptomatic, severe tricuspid regurgitation (TR) undergoing TTVR in the first year or so after Evoque’s U.S. commercial debut.
“Rates of new CIED implantation and bleeding were lower than randomized clinical trial experience,” Raj Makkar, MD, of Cedars-Sinai Medical Center in Los Angeles, and colleagues reported in JAMA. “Taken together, the findings suggest that introduction of TTVR after regulatory approval in the U.S. has been safe and effective.”
Concerns about TTVR resulting in excess bleeding and conduction abnormalities had emerged in the TRISCEND II trial, the study that was the basis for Evoque’s FDA approval in February 2024.
In that randomized trial, Evoque TTVR was deemed superior to medical therapy based on a win ratio analysis with the difference driven primarily by improvements in symptoms and quality of life. However, the tradeoffs were more severe bleeding (15.4% vs 5.3%, P=0.003) and new permanent pacemakers (17.4% vs 2.3%, P<0.001) compared with medical therapy alone as the control.
TRISCEND II nevertheless sufficed for regulators to approve the option for the typically older, relatively sick TR patients, who have historically had few treatment options outside surgery.
Makkar and colleagues remarked on the bleeding rate being brought down to 7.9% in the real world.
“In TRISCEND II, postprocedural anticoagulation plus antiplatelet therapy was recommended in all patients up to 6 months, compared with approximately 40% of patients discharged with combined anticoagulation and antiplatelet therapy in the STS/ACC TVT Registry, likely contributing to the lower severe bleeding rates reported in this study,” they surmised.
Meanwhile, late-onset conduction abnormalities remain a concern with TTVR due to radial expansion of the device and compression of the AV node.
“Furthermore, TTVR may be more challenging in patients with a preexisting CIED because it can impair visualization and complicate valve deployment, and data regarding procedural performance and outcomes in patients with vs without baseline CIEDs are limited,” Makkar and colleagues acknowledged.
The Evoque system features a trileaflet bovine pericardial valve, mounted in a self-expanding nitinol frame with an intra-annular fabric sealing skirt, that is delivered via transfemoral venous access. TTVR procedures are typically performed under general anesthesia with multimodality imaging.
There is much competition on the horizon, with several investigational TTVR devices moving through the pipeline. Another transcatheter technology for TR, the TriClip G4 transcatheter edge-to-edge repair (T-TEER) device, is already FDA approved for improving quality of life and functional status.
In the end, the choice of technology depends on the patient, according to Makkar’s group, as randomized head-to-head comparisons are still lacking.
“Utilization of TTVR may have a potential advantage over T-TEER in patients with large coaptation gaps, significant leaflet tethering, or preexisting transvenous leads. These complementary profiles support a pragmatic selection framework: consider T-TEER when residual TR is acceptable and pacing risk should be minimized, and consider TTVR when the therapeutic goal is near elimination of TR or anatomy is unfavorable for repair,” the group wrote.
Makkar and colleagues’ retrospective cohort study included all consecutive patients who underwent TTVR in the U.S. from February 2024 to March 2025. These were patients with symptomatic, severe TR despite optimal medical therapy, for whom 1,034 TTVR procedures were attempted at 82 centers.
The cohort averaged 77.1 years of age, and 69.1% were women. Nearly three in four were in New York Heart Association (NYHA) functional class III/IV. The mean left ventricular ejection fraction was 55.9%. Additionally, 83.8% had atrial fibrillation or flutter.
TR was confirmed to be severe or greater in the vast majority of participants (96.7%). The etiology of TR was judged to be primary (41.2%), secondary (43.2%), pacemaker-induced (9.4%), or other (6.1%). About 70% of patients were considered at high surgical risk; another 10.5% were deemed extreme-risk. Moderate or severe mitral regurgitation was also present in 39.9% of patients.
Ultimately, the Evoque valve was successfully implanted in 98.4% of cases. Mild or less TR was achieved in 98.4% of patients post-TTVR (97.7% at 30 days).
Thirty-day outcome rates were:
- All-cause mortality: 3.1%
- Stroke: 0.2%
- Bleeding: 7.9%
- New cardiac implantable electronic device (CIED): 10.1% overall and 15.9% in CIED-naive patients
- Heart failure hospitalization: 3.1%
As expected, patients reported clinically meaningful improvements in health status and quality of life from baseline to 30 days post-implantation. NYHA functional class improved to I/II in 82.7% of Evoque recipients; Kansas City Cardiomyopathy Questionnaire Overall Summary scores reached 22.4 points.
The 38.0% of patients with a CIED at baseline did not have significantly worse 30-day mortality, heart failure hospitalization, and functional outcomes, according to Makkar’s group. “Procedural duration and TR reduction were comparable regardless of CIED presence, bleeding and mortality rates remained low, and health status improved substantially across both groups, reinforcing the applicability of TTVR across a broad patient population.”
Importantly, the study authors acknowledged the observational nature of their study and the results being limited to the first 30 days.
Source link : https://www.medpagetoday.com/cardiology/interventionalcardiology/120771
Author :
Publish date : 2026-04-13 19:56:00
Copyright for syndicated content belongs to the linked Source.