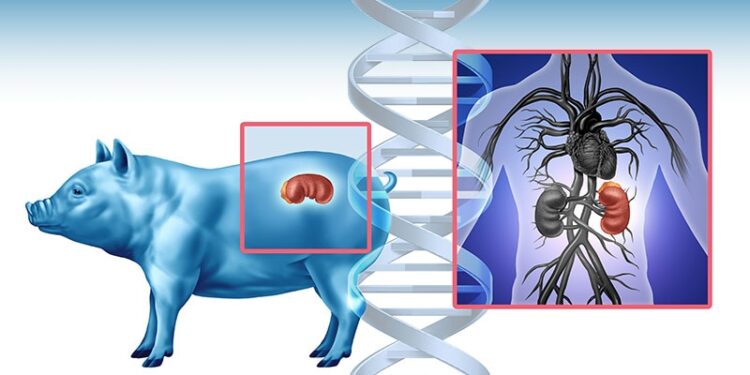
Nearly a year after performing the first transplant of a pig kidney into a living human recipient, Massachusetts General Hospital (MGH), Boston, reported that a second patient is doing well after a similar procedure, while the field takes a big step forward with a new approval from the US Food and Drug Administration (FDA) for clinical trials.
“This second xenotransplant provides us with another excellent opportunity to learn how we can make genetically edited pig organs a viable, long-term solution for patients,” said surgeon Tatsuo Kawai, MD, PhD, director of the MGH’s Legorreta Center for Clinical Transplant Tolerance, Boston, in a press statement.
“Although we have a long way to go to make that a reality, this transplant is an important next step that has given us optimism to achieve that goal,” he said.
MGH reported its second pig-to-human kidney transplant was completed in January in a 66-year-old man, identified as Tim Andrews of Concord, New Hampshire, with end-stage renal disease (ESRD), who had been on dialysis for more than 2 years.
In addition to having complications, including a recent heart attack, Andrews had the less common O− group blood type, which can double the amount of time patients spend on the kidney waiting list, up to 10 years, MGH reported.
Using CRISPR-Cas9 technology, scientists altered a donated pig kidney with 69 genomic edits to remove potentially harmful pig genes and to add human genes to improve compatibility.
The team also took measures to inactivate endogenous porcine retroviruses in the donated kidney to eliminate infection risk, and novel immunosuppressant medications were employed.
The 2.5-hour transplant procedure was successfully performed on January 25 and Andrews was discharged on February 1.
MGH reported that the patient is “off dialysis for the first time in over 2 years, with his new kidney functioning as expected.”
Andrews is the fourth living human to have received a kidney transplant from a porcine donor; the second to do so at MGH. The first MGH patient survived for 52 days.
Of the two other cases, one involved a 54-year-old woman who received a pig’s kidney and a heart pump on different days in April 2024. Following a decline, the failing kidney had to be removed after 47 days. The patient survived for 86 days following the transplant.
In the other case, performed at NYU Langone Transplant Institute in New York City, a 53-year-old woman received a pig’s kidney, modified with 10 gene edits, in November. She is currently the longest living recipient of a pig organ transplant.
The patient, at day 84 post-transplant at press time, was “doing great,” Jeffrey M. Stern, MD, a member of the NYU transplant team, told Medscape Medical News.
He noted that the patient, who had been on kidney dialysis for nearly 9 years, had donated a kidney to her mother in 2001, and after developing preeclampsia in pregnancy developed other complications.
“Despite being at the top of the waiting list for most of those years, the patient had received only one kidney offer, which turned out to not be a match,” Stern said. “She really had no other options, and was considered an ideal candidate for xenotransplantation [the transplant of organs from one species to another],” he said.
FDA Approves First Clinical Trials
The pig kidney transplant cases conducted so far were permitted under the FDA’s Expanded Access Protocol or compassionate use program, which allows investigational procedures outside of clinical trials only when patients are critically ill and running out of options.
However, United Therapeutics Corp., which provided the pig donor kidney for the case at NYU Langone Transplant Institute, reported in a press statement that it has received FDA approval to conduct the first clinical trials involving pig donated kidney transplants.
The trial will involve a cohort of six patients with ESRD who will receive an investigational “UKidney” derived from a 10 gene–edited source pig.
To be considered for inclusion, patients must be aged between 55 and 70 years and have been on dialysis for at least 6 months.
In addition, trial candidates will include “ESRD patients who have been assessed and determined to be ineligible for a conventional allogeneic kidney transplant for medical reasons, and ESRD patients who have been on the kidney transplant waitlist but are more likely to die or go untransplanted than receive a deceased donor kidney transplant within 5 years,” the company reported.
After transplantation, patients will be monitored for 24 weeks, and decisions on the expansion of the study will be made after an independent review. The first trial is expected to begin in mid-2025.
After the initial cohort, the trial is expected to expand to up to 50 patients. All patients receiving the transplant will continue to be followed for the rest of their lives, with monitoring of kidney function, zoonotic infections, and survival.
Previous Skepticism Shifts to Optimism
In an editorial addressing future prospects and challenges in xenotransplantation, Stern and his coauthors underscore that, as the first in-human clinical trials draw near, “critical clinical challenges include determining the optimal donor genetic constructs and immunosuppressive regimens.”
They added that complexities surround enrollment criteria and patient selection, in addition to “ethical concerns such as lifelong zoonosis monitoring.”
“Only a limited number of centers have the expertise needed to conduct these complex trials,” they added.
Nevertheless, after years of speculation about the viability of xenotransplantation, Stern said he is optimistic about the current prospects.
“There was [a quote] that ‘xenotransplantation is the future — and always will be,’ and for a long time, everyone thought that, and even I thought that about a decade ago,” he told Medscape Medical News.
“But I am much more optimistic right now that this is a viable solution on the horizon for some patients,” he said.
“After the clinical trials are performed, and with a few more advancements, I think this will hopefully be an everyday practice to alleviate the need for organs on the waiting list.”
According to the American Kidney Fund, more than 106,000 people are on the national list for kidney transplants in the United States, yet only about 27,000 such transplants were performed in 2023. Meanwhile, as many as 557,000 patients are on dialysis.
Michael Curtis, PhD, CEO of eGenesis, Cambridge, Massachusetts, which provided the genetically edited pig kidney for the MGH transplants, underscored the significance of the latest transplant in light of those figures.
“This procedure is more than a scientific milestone — it represents a new frontier in medicine,” he said in a press statement. “We stand at the beginning of a future where organ shortages may no longer dictate patient outcomes.”
Stern reported having no disclosures.
Source link : https://www.medscape.com/viewarticle/second-pig-kidney-transplant-success-and-fda-trial-approval-2025a1000819?src=rss
Author :
Publish date : 2025-04-03 13:29:00
Copyright for syndicated content belongs to the linked Source.