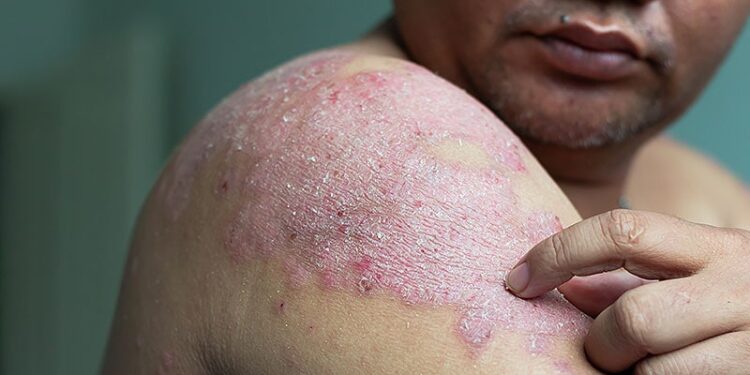
ORLANDO, Fla. — In moderate to severe plaque psoriasis, two highly selective oral TYK2 inhibitors produced a level of response in phase 2 studies suggesting that drugs in this class might rival biologics.
In one, a 75% skin clearance on the Psoriasis Area Severity Index (PASI) was achieved with a drug currently known as ICP-488 in nearly 80% of patients by week 12. In the other, an open-label extension (OLE) trial of a previously reported 12-week STRIDE trial with a drug known as ESK-001, more than 60% of patients on the most effective dose achieved 90% clearance (PASI90) by 52 weeks.
Both of the studies were dose-ranging, but the highest and more effective doses produced response rates comparable to those previously reported with biologics in studies of comparable duration. Each study was presented as a late-breaker at the American Academy of Dermatology (AAD) 2025 Annual Meeting.
Nearly 80% Achieve PASI75 Response at 12 Weeks
In the double-blind study with ICP-488, which randomized 129 patients in a 1:1:1 ratio to placebo or either 6 mg or 9 mg of the active agent administered once daily, the PASI75 response in the highest dose group was 78.6% at the end of 12 weeks. The PASI75 response with the 6-mg dose at 77.3% was only slightly lower, and both were nearly eightfold greater (P < .0001) than the 11.6% response in the placebo group.
The PASI90 response at the end of 12 weeks was 50.0% in the 9 mg once-daily arm, reported Xiaoguang Zhang, MD, PhD, a senior clinician and researcher at Hebei University, Baoding, China. This was considerably higher than the 36.5% PASI90 response in the 6 mg once-daily arm, but the response was 0% in the placebo arm (P < .0001 vs both doses of ICP-448).
The same types of differences between active treatment and placebo were seen for the Static Physician’s Global Assessment (sPGA) scores of 0 (clear) and 1 (almost clear). The proportion of patients achieving an sPGA score of 0/1 at 12 weeks was 71.4% and 70.5% for the 9-mg and 6-mg doses of ICP-488, respectively, vs 9.3% for placebo (P < .0001 vs both doses of ICP-448).
The drug was well tolerated. According to Zhang, “all treatment-emergent adverse events were mild to moderate, and no patient discontinued treatment due to adverse events.”
As a once-daily oral therapy, based on the data from this study, ICP-488 will be “a valuable treatment option for moderate to severe plaque psoriasis” if phase 3 data confirm these findings, Zhang said.
ICP-488 and ESK-001 Perform Similarly at Week 12
The 12-week results with the 9-mg dose of ICP-488 were similar to the 12-week results in the previously reported but not yet published STRIDE trial of ESK-001. In the 12-week data reported at the 2024 AAD meeting, 64.1% of those randomized to the most effective dose, which was 40 mg twice daily, achieved the primary PASI75 endpoint. For those randomized to the 40-mg once-daily dose, the proportion was 56.4%.
In the newly presented 52-week STRIDE OLE study of ESK-001, patients were re-randomized after the 12-week efficacy analysis and an additional 4-week safety analysis, according to the principal investigator, Andrew Blauvelt, MD, a practicing dermatologist and president of the Oregon Medical Research Center, Portland, Oregon.
So 16 weeks after initial enrollment, all groups, including those initially in the placebo arm, began the 52-week OLE on either a 40 mg once-daily or twice-daily dose of ESK-001. However, the protocol was amended at week 28 of the OLE because it had become apparent that a twice-daily dose was more active but equally well tolerated. The analysis at 52 weeks evaluated those who started STRIDE on 40 mg twice daily and remained on this dose for the length of the OLE, those who first started on 40 mg twice daily at the beginning of the OLE, and those who started on the 40 mg once-daily dose but were switched to the twice-daily dose 28 weeks into the OLE, Blauvelt explained.
50% of Patients Previously Exposed to Biologics or Janus kinase inhibitor (JAKi)
About 50% of the 165 patients enrolled in the OLE study had received biologics or non-TYK2 JAKis previously. Their median age was nearly 50 years, and 27 (16%) had been assigned to 40 mg twice daily in STRIDE and therefore received this dose continuously for 52 weeks.
When evaluating responses specifically in the group on continuous 40 mg twice daily of ESK-001 for the entire 52 weeks, skin clearing generally improved over time. For example, PASI75 rates peaked at 82.7% at 28 weeks before plateauing. At week 52, 77.5% retained this level of response. PASI90 rates reached 63% at week 28 and remained near that level (61.3%) at week 52. PASI100 rates climbed to 30.9% at week 28 and reached 38.8% by week 52.
Of those originally assigned to 40 mg twice daily, “62% showed continued improvement in PASI response at week 52 compared to week 12,” reported Blauvelt, who said that the persistent improvement over time is consistent with experimental studies with TYK2 inhibitors.
“TYK2 is a member of the JAK family but a unique member and pretty distinct from JAK1, JAK2, and JAK3,” Blauvelt explained.
One distinction is that TYK2 inhibition is more closely associated with inhibiting cytokines upregulated in patients with psoriasis, such as interleukin (IL)-23, IL-17A, and IL-17F. Another is that drugs highly selected for TYK2 do not appear to produce the risks, such as cardiovascular adverse events, which are included in boxed warnings for JAKis.
In the 12-week STRIDE trial, 58.5% of patients treated with 40 mg twice-daily ESK-001 achieved a Dermatology Life Quality Index score of 0 or 1, which indicates no effect on a patient’s life related to the skin condition. At the end of week 52 in the OLE, the proportion was 61.3%. Similarly, 73.2% achieved an average Numerical Rating Scale pruritus score < 4, which indicates no greater than moderate itch. The proportion at week 52 of the OLD was 81.3%.
Like the experience with ICP-488, the majority of treatment-emergent adverse events were mild to moderate in the STRIDE OLE. According to Blauvelt, there are no significant safety signals to date.
Driven by these efficacy and safety results, the ONWARD study, which is a phase 3 trial evaluating ESK-001 for regulatory approval, has already enrolled more than 600 patients. According to Blauvelt, given the inability of biologics to provide adequate control to many patients with moderate to severe psoriasis, “new therapeutic options are needed,” and TYK2 inhibitors appear particularly promising.
Zhang reported no potential conflicts of interest. Blauvelt reported financial relationships with more than 25 pharmaceutical companies including Alumis, which provided funding for the STRIDE trial.
Source link : https://www.medscape.com/viewarticle/two-oral-tyk2-inhibitors-enter-phase-3-trials-plaque-2025a100081l?src=rss
Author :
Publish date : 2025-04-03 12:06:00
Copyright for syndicated content belongs to the linked Source.