Note that some links may require registration or subscription.
An interventional cardiologist who was fired filed a lawsuit against UPMC, alleging retaliation and violation of Pennsylvania’s Whistleblower Act after he reported colleagues’ racist text messages and the CEO’s conflict of interest. (PennLive)
Researchers funded by the Advanced Research Projects Agency for Health (ARPA-H) of HHS regenerated cartilage and bone in osteoarthritic animal models, moving a step closer to human clinical trials, the agency announced.
Since the Trump administration began overhauling foreign aid last year, U.S.-based organizations received huge infusions of cash while smaller groups in other nations didn’t — the opposite of what Trump promised. (New York Times)
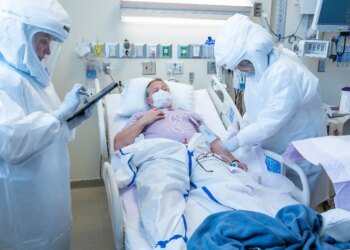
The family of a young ICU patient in Connecticut who died while in the care of a teledoctor sued the hospital, accusing it of negligence. (NBC News)
A CMS pilot initiative allows some providers to offer hemp-based products to patients when clinically indicated, excluding inhalable products. (Fierce Healthcare)
Abbvie and Genentech became the latest companies to provide prescription drugs at a reduced rate on the TrumpRx site. (CBS News)
Resident doctors in England started a 6-day walkout today after rejecting an offer the government said would not get better. (Reuters)
Medical supplies and therapeutic foods are stuck in Dubai as a result of the war in Iran, leaving humanitarian centers and clinics across the world without resources. (NPR)
After Idaho cut services for people with schizophrenia, some patients died. (New York Times)
Respiratory syncytial virus infections have continued later this year, prompting many states to extend the immunization period for eligible infants and toddlers to the end of April. (CNN)
The U.S. reported 98 new measles cases in the past week, according to the Yale School of Public Health. Here’s where they were.
A measles outbreak has killed more than 100 children in Bangladesh in less than a month, sparking an emergency vaccination campaign. (AP)
Colorado enacted the nation’s first law banning arrests solely based on colorimetric drug tests, which researchers said have high false positive rates. (CNN)
ImmunityBio said it will have more stringent protocols for promotional communications in response to an FDA warning letter saying the company made “false or misleading” claims about its bladder cancer drug, nogapendekin alfa inbakicept (Anktiva).
Source link : https://www.medpagetoday.com/cardiology/interventionalcardiology/120670
Author :
Publish date : 2026-04-07 13:44:00
Copyright for syndicated content belongs to the linked Source.