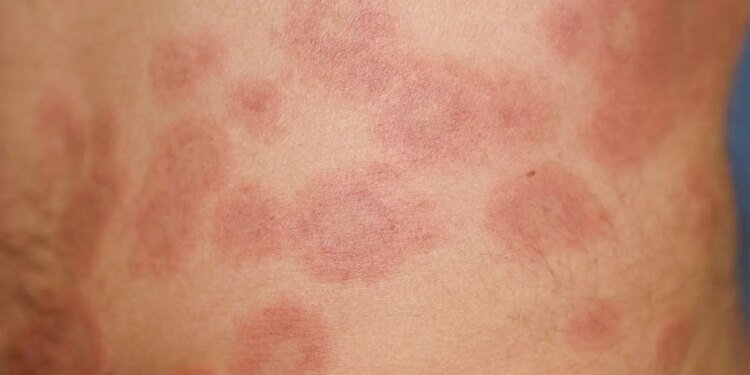
- Cutaneous lupus erythematosus (CLE), which has no specific approved therapies, is believed to be driven by excessive interferon and B-cell activity, which in turn are triggered by Toll-like receptors (TLR) 7 and 8.
- Enpatoran is a small-molecule oral agent that inhibits TLR 7 and 8.
- In this part of a phase II trial of enpatoran, the drug was superior to placebo in relieving lupus skin manifestations.
Enpatoran, an investigational oral drug that inhibits Toll-like receptors (TLR) 7 and 8, was superior to placebo in relieving lupus skin manifestations in a dose-finding phase II trial.
Doses of 25, 50, and 100 mg twice daily reduced Cutaneous Lupus Disease Area and Severity Index-Activity (CLASI-A) scores by 64-72 percentage points from baseline by week 16, versus a 44 percentage point decrease with placebo (all P<0.05 vs placebo), according to Eric Morand, MBBS, PhD, of Monash University in Melbourne, Australia, and colleagues. The publication in The Lancet Rheumatology confirms data first reported last year at a lupus conference.
Each step-up in dosing in the trial, dubbed WILLOW, provided 4 additional percentage points in the CLASI-A reduction. The investigators’ statistical analysis showed a significant dose-response relationship (P<0.0002).
But in an accompanying commentary, Cristina Arriens, MD, of the Oklahoma Medical Research Foundation in Oklahoma City, wasn’t buying it.
The confidence intervals around the CLASI-A reductions at each dosage level overlapped considerably, leading Arriens to state that the “data were insufficient to clearly differentiate between the doses.” Moreover, she wrote, an illustration in the paper “suggests that the highest dose (100 mg) might not have yielded the best response.”
This report follows another meeting presentation and subsequent journal paper about a separate portion of the phase II trial, in which the three enpatoran doses were tested in patients with systemic lupus erythematosus (SLE) with or without skin involvement (called Cohort B). In that analysis, no dose-response relationship could be seen, except perhaps a negative one (i.e., the lowest dose produced the largest clinical benefit).
Still, it seemed clear that enpatoran was clinically effective enough to warrant phase III testing, which is now underway in two identical placebo-controlled trials, ELOWEN-1 and ELOWEN-2, with dosing at 50 mg twice daily.
TLRs have drawn attention as a potential treatment because these components of the innate immune system appear to play a strong role in lupus by triggering interferon release and B-cell activation. Enpatoran is a small-molecule agent that inhibits TLR 7/8 and performed as hoped in preclinical models.
While Cohort B enrolled patients with SLE and evaluated outcomes with standard lupus measures, such as so-called BICLA response, the current report focused on Cohort A, in which participants had to have substantial skin involvement. This could be a formal cutaneous lupus erythematosus (CLE) diagnosis, or SLE with mild or absent non-mucocutaneous symptoms but baseline CLASI-A scores of at least 8. Around 55% of the cohort had CLE.
Cohort A enrolled 102 patients who were randomized in nearly equal numbers to placebo or one of the three enpatoran doses. Median age in the four groups ranged from 41 to 49; roughly three-quarters were women. Just under half were white, about 12% were American Indian or Alaska native, about 30% were Asian, and the rest were Black. Close to 40% of the entire cohort reported Hispanic ethnicity. This distribution reflected the trial’s international character, with enrollment about equally divided among Asia, South or Central America, and North America and Europe.
CLASI-A scores, which formed the primary outcome measure, averaged 12-16 on the 70-point scale in the four groups at baseline. Secondary outcomes included investigators’ global assessments, fatigue scores, and Skindex quality of life measures.
While Morand and colleagues claimed a dose-response relationship for CLASI-A scores, there was no hint of it for the secondary outcomes. For many, the 100-mg dose showed the least effect, although generally still significantly superior to placebo.
On the other hand, one could see a potential dose-response for adverse effects. Grade 3-4 adverse events occurred in 8% of the 100-mg group compared with 4% with the two lower dosages. Similarly, 8% of the high-dose group stopped the drug because of adverse events, versus 0-4% of the placebo and lower-dosage groups.
It’s worth emphasizing, though, that with around 25 participants per group, 8% equates to just two participants. The phase III trials, each with about 200 participants, should give a better sense of the drug’s safety. In WILLOW, the most common adverse effect was upper respiratory infections, seen in five members of the 100-mg group, four assigned to 50 mg, and two each in the 25-mg and placebo groups. Diarrhea, urinary tract infections, herpes zoster activations, and liver enzyme elevations also occurred and with a similar pattern — but each also involved just one or two patients per group. Morand and colleagues called enpatoran “well tolerated” on the basis of these data.
Source link : https://www.medpagetoday.com/rheumatology/lupus/121208
Author :
Publish date : 2026-05-11 18:40:00
Copyright for syndicated content belongs to the linked Source.