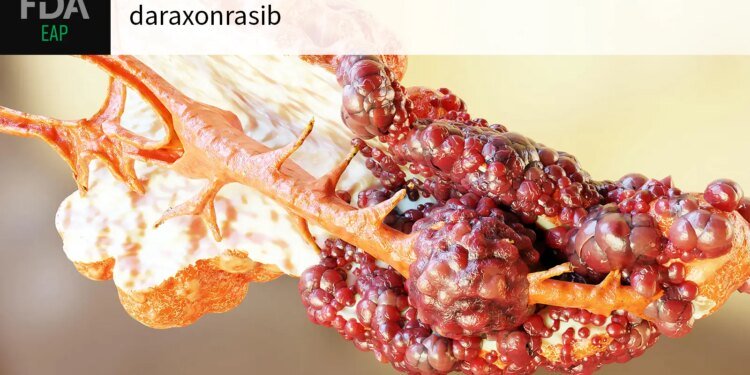
The FDA has given the green light to an expanded access program (EAP) for the investigational pancreatic cancer drug daraxonrasib.
In a “safe to proceed” letter to Revolution Medicines, the FDA gave its approval to initiate the EAP in collaboration with licensed prescribers. The approval came just 2 days after the FDA received the company’s application for an EAP.
“Granting the request 2 days after receiving the expanded access application reflects the FDA’s strong commitment to facilitate early access to therapies for serious and life-threatening conditions, including pancreatic cancer,” FDA Commissioner Marty Makary, MD, MPH, said in a statement. “Having taken care of many patients with metastatic cancer, I am hopeful that today’s action will improve the lives of patients suffering from this disease.”
The EAP approval for daraxonrasib continues a string of good news that began just 3 weeks ago when the company announced an “unprecedented” improvement in overall survival with the RAS inhibitor in patients with previously treated metastatic pancreatic cancer. Median OS essentially doubled from 6.7 months with standard therapy to 13.2 months with daraxonrasib. A week later two small studies reported at the American Association for Cancer Research meeting showed response rates of about 50-60% and disease control in 90-92% of patients with previously untreated metastatic pancreatic cancer who received the RAS inhibitor as a single agent or in combination with chemotherapy.
According to the company announcement, details of the study in previously treated metastatic disease will be reported at the American Society of Clinical Oncology meeting at the end of May.
The drug is also generating buzz because former Sen. Ben Sasse (R-Neb.), diagnosed with pancreatic cancer in December, has recently talked about his participation in a clinical trial of daraxonrasib, saying the drug shrank his tumor by 76%.
An EAP provides a means to extend access to promising investigational drugs to more patients treated under a specific protocol or as part of investigational new drug application. Requests for expanded access must be submitted to the sponsor by U.S.-licensed physicians on behalf of eligible patients.
Source link : https://www.medpagetoday.com/hematologyoncology/othercancers/121081
Author :
Publish date : 2026-05-01 19:30:00
Copyright for syndicated content belongs to the linked Source.