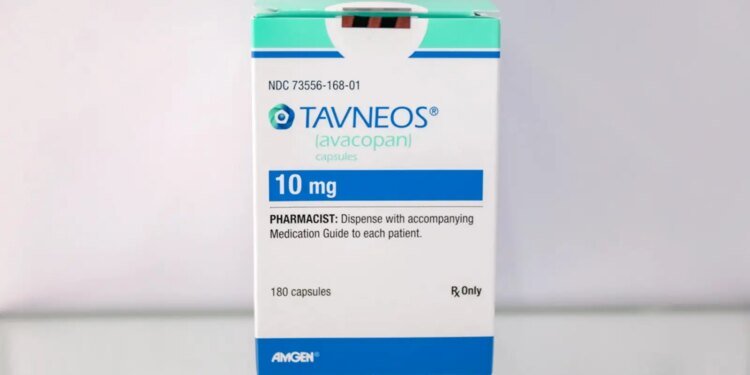
The FDA on Tuesday warned that postmarketing data has turned up dozens of serious cases of drug-induced liver injury (DILI) associated with avacopan (Tavneos), a vasculitis drug that the agency wants removed from the market.
Eight of the cases were fatal, including three patients who developed vanishing bile duct syndrome (VBDS), a condition that can lead to permanent liver damage where the bile ducts are progressively destroyed before disappearing altogether.
“Although hepatotoxicity is a serious adverse reaction for Tavneos identified in premarket clinical trials and described in product labeling, VBDS and DILI cases with fatal outcomes represent new safety concerns,” the FDA said.
Avacopan was approved in 2021 as an adjunctive treatment for adults with severe anti-neutrophil cytoplasmic autoantibody (ANCA)-associated vasculitis (granulomatosis with polyangiitis and microscopic polyangiitis [GPA and MPA]). The oral complement 5a inhibitor is typically used in combination with glucocorticoids and other standard medications.
FDA’s review of postmarketing data globally through Oct. 9, 2024 turned up 76 DILI cases with enough evidence to suggest a causal link to avacopan, including 54 hospitalizations and five U.S. cases. Median time to DILI onset was 46 days (range 22-140).
Seven biopsy-confirmed VBDS cases were reported, all requiring hospitalization. Initial signs and symptoms of VBDS often include jaundice, fatigue, and pruritus.
The pattern of liver injury for 38 of the DILI cases was cholestatic or mixed, often with substantial elevations in alkaline phosphatase (ALP) and total bilirubin, while the pattern in the VBDS cases was cholestatic or mixed for four patients and hepatocellular for three patients.
According to the FDA, physicians treating with avacopan should conduct regular liver panel testing and discontinue if patients develop symptomatic cholestasis or if liver enzymes spike to over three times the upper limit of normal (ULN) for alanine aminotransferase or aspartate aminotransferase or over two times the ULN for ALP.
“If liver test abnormalities or symptoms of liver injury do not improve, patients should be referred to a hepatologist for further evaluation,” the agency said.
The FDA in January requested that avacopan be voluntarily withdrawn from the market, raising concerns about cases of liver injury as well as the trial data underpinning its approval, but drugmaker Amgen rebuffed the agency’s request.
“Amgen remains confident that Tavneos represents an important treatment option for patients with severe, active ANCA-associated vasculitis (GPA and MPA), rare, serious autoimmune diseases with limited therapeutic alternatives,” the company said in a letter to healthcare providers at the time.
Avacopan was approved primarily off findings from ADVOCATE, which randomized patients to either avacopan or prednisone tapering with standard therapy in each arm (cyclophosphamide or rituximab).
In the phase III study, remission at week 26 was observed in 72.3% of the avacopan group and in 70.1% of the prednisone group, meeting criteria for noninferiority. Sustained remission at week 52 was observed in 65.7% of the avacopan group and in 54.9% of the prednisone group, which was significant for both noninferiority and superiority.
Serious adverse events related to liver-function testing occurred in 5.4% of patients in the avacopan group and in 3.7% of those in the prednisone group.
Source link : https://www.medpagetoday.com/rheumatology/generalrheumatology/120577
Author :
Publish date : 2026-03-31 17:51:00
Copyright for syndicated content belongs to the linked Source.