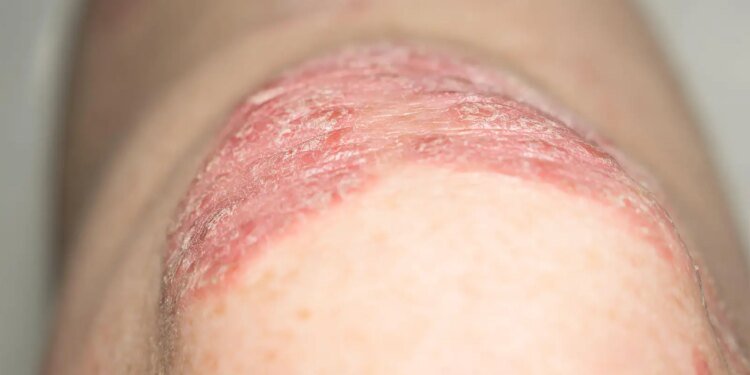
- Available evidence suggests GLP-1 receptor agonists warrant consideration as an adjunct to conventional treatments for psoriasis.
- Multiple studies have shown that GLP-1 receptor agonists improve psoriasis disease status and quality of life.
- The greatest benefits occur in patients with obesity, as weight loss appears to enhance effects of psoriasis medications.
GLP-1 receptor agonists represent a “biologically plausible and clinically intriguing adjunct” to psoriasis therapy for selected patients, authors of a National Psoriasis Foundation (NPF) “primer” concluded.
The evidence showed that GLP-1 agonists and dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 agents have been associated with clinically meaningful reductions in the Psoriasis Area and Severity Index (PASI), accompanied by improvements in quality of life. Additionally, semaglutide (Ozempic, Wegovy) and liraglutide (Victoza, Saxenda) have been associated with reductions in C-reactive protein, interleukin-6, lipids, and visceral adiposity.
Small translational studies have shown correlations between improvement in PASI scores and reductions in superficial adiposity and dermal γδ T-cell density. GLP-1 receptor agonists can be combined safely with methotrexate, cyclosporine, and biological agents used to treat psoriatic diseases. Preliminary data have shown both metabolic and immunomodulatory benefits, the authors stated in JAMA Dermatology.
“We think the paper is important because it’s actually addressing the question of whether dermatologists and dermatology providers should be prescribing the drugs,” study author Andrew Blauvelt, MD, of Blauvelt Consulting in Annapolis, Maryland, told MedPage Today. “It’s more about how to prescribe them, giving the names of the drugs, the doses, the expected side effects, the types of follow-up needed. It’s more than a review paper, so we call it a primer. It’s quite extensive and comprehensive.”
“[Clinicians] are prescribing them for weight loss, and we don’t need more data on that,” he added. “Weight loss, in the case of psoriasis, will improve the ability of our psoriasis drugs to work.”
The available evidence has made a case for considering adjunctive use of GLP-1 agonists in selected patients with psoriasis and metabolic comorbidities, Blauvelt and colleagues noted. However, many studies have involved small numbers of patients, were of short duration, and lacked a control group. Definitive conclusions require larger, randomized clinical trials.
More compelling data have already begun to emerge. At the recent American Academy of Dermatology (AAD) meeting, the first report from the TOGETHER-PsA trial showed that adding tirzepatide (Zepbound) to ixekizumab (Taltz) significantly increased the number of patients with psoriatic arthritis (PsA) who met the dual endpoint of at least 50% improvement from baseline in American College of Rheumatology criteria and at least 10% weight loss, as compared with ixekizumab monotherapy.
An ongoing companion study (TOGETHER-PsO) is comparing the combination and single-agent ixekizumab in patients with moderate/severe psoriasis. Offering a sneak peek at the data during his AAD presentation, Joseph Merola, MD, of UT Southwestern Medical Center in Dallas, said more patients randomized to the combination have achieved PASI goals (PASI 75, PASI 90, and PASI 100).
According to the NPF primer, GLP-1 receptor agonists are associated with PASI reductions in the range of 40-80% in patients who have psoriasis and comorbid obesity or type 2 diabetes. Data from adequately sized cohorts show improvements of 48-52%. The efficacy may depend on a patient’s degree of metabolic dysfunction at baseline. Three randomized trials of GLP-1 receptor agonists in patients with psoriasis and concurrent obesity and/or type 2 diabetes have produced mixed results. In a placebo-controlled trial involving glucose-tolerant patients with psoriasis, GLP-1 receptor agonists did not significantly affect psoriasis status.
Evidence reviewed for the primer showed that GLP-1 agonists target shared metabolic and inflammatory pathways in psoriasis. A majority of patients with psoriasis have concurrent overweight/obesity, with some estimates ranging as high as 75-80%. In the absence of overweight/obesity, the impact of GLP-1 receptor agonists appears modest.
“There is a little bit of data that we discussed in the paper on the benefit of GLP-1s as an anti-psoriatic effect,” said Blauvelt, who also chairs the NPF medical board. “We believe that the majority of the benefits are going to be coming from the weight loss, which are making the psoriasis drugs work better and not independent psoriatic activity of the drugs. There might be little bit, but we don’t think it’s a lot.”
A study unavailable during the NPF evidence review added more information for the discussion about mechanisms of action. Investigators evaluated genetic proxies of GLP-1 receptor expression in blood samples from 36,466 patients with psoriasis (458,078 controls) and 5,065 patients with psoriatic arthritis (21,286 controls). As a positive control, they also examined associations between genetic proxies of GLP-1 receptor expression and cardiometabolic outcomes with the known therapeutic responsiveness to GLP-1 receptor agonists.
After adjustment for genetic proxies of metabolic traits, the results showed that genetic proxies of increasing GLP-1 receptor expression were associated with reduced susceptibility to psoriasis (OR 0.72, 95% CI 0.68-0.77) and to psoriatic arthritis (OR 0.48, 95% CI 0.40-0.58). The associations remained statistically significant after adjustment for metabolic traits.
“The present genetic analyses suggest that increasing GLP-1 receptor expression is associated with reduced susceptibility to psoriasis and PsA, at least partly independent of metabolic traits, supporting further investigation of non-metabolic mechanisms,” the investigators concluded.
Source link : https://www.medpagetoday.com/dermatology/psoriasis/121079
Author :
Publish date : 2026-05-03 14:00:00
Copyright for syndicated content belongs to the linked Source.