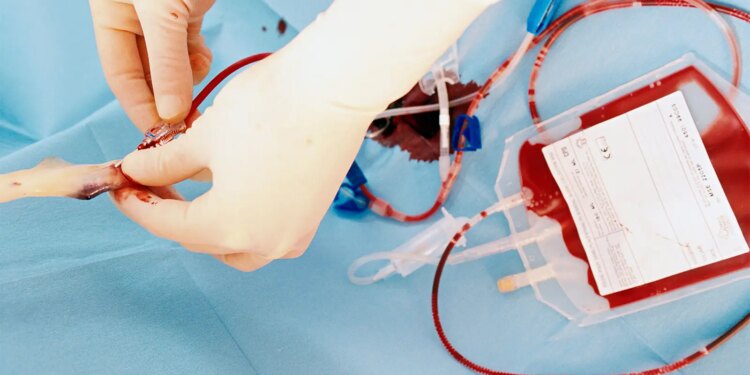
- Adding a pooled umbilical cord blood product to HLA-matched single-unit cord blood transplants resulted in 96% disease-free survival in acute leukemia and other hematologic malignancies.
- No grade 3 or 4 severe graft-versus-host disease occurred in patients who received the pooled cord blood product.
- The off-the-shelf product addresses several limitations of single-unit cord blood transplants.
Stem cell transplants for acute leukemia using pooled, non-human leukocyte antigen (HLA)-matched cord blood led to near-perfect engraftment and no severe graft-versus-host disease (GVHD) in a small safety-focused study.
After a median follow-up of 1.4 years, 27 of 28 patients remained alive and disease free after transplants with dilanubicel, a cryopreserved expanded progenitor cell product, added to a single matched cord blood unit. No grade 3/4 acute or chronic GVHD occurred in any patient.
The results have provided the basis for prospective, multicenter studies to evaluate dilanubicel as an adjunct to different types of allogeneic hematopoietic cell transplant platforms, including matched and haploidentical donors, reported Filippo Milano, MD, PhD, of Fred Hutch Cancer Center in Seattle, and colleagues in the Journal of Clinical Oncology.
“It’s quite fascinating, because basically, we give one unit that matches with the patient,” Milano told MedPage Today. “Then we pair with this other unit, a pooled unit, which usually contains between six and eight cord blood units pulled together, so we are basically doing a transplant with up to nine different individuals.”
“The matched unit sees all these different mismatches in the [pooled unit] and reacts to it,” he added. “Every patient enrolled in the study, around day 7 or day 10, has spiking lymphocytes that are coming from the matched unit, reacting to the pooled unit. We think this has led to a very potent effect in controlling the leukemia and producing very good overall survival. But one of the most important findings is the lack of grade 3 and 4 acute graft-versus-host disease.”
They addressed one of the key limitations of umbilical cord transplants — the finite cell dose.
“There are side effects that come with a limiting cell dose, there is an impediment to engraftment,” said Jeffery Auletta, MD, of the National Marrow Donor Program in Minneapolis. “This study is encouraging because it shows that this adjuvant cell therapy can help engraftment of a single umbilical cord blood unit.”
The results require confirmation in a larger, multicenter study to ensure that the findings are generalizable beyond a single center, he added. A second consideration is to work out details of manufacturing and distribution for dilanubicel.
“It seems like it’s a readily available product because it’s off the shelf, but those regulatory details will have to be worked out from a logistics standpoint too,” said Auletta.
Cord blood transplantation offers a potential cure for hematologic malignancies, particularly for patients without matching donors, and has been associated with a reduced risk of relapse in high-risk disease. However, the transplants carry a higher risk of delayed engraftment and immune recovery, as well as increased early treatment-related morbidity. To overcome limitations, double cord blood unit transplantation has been adopted in many instances. However, the approach costs more and has an increased risk of severe acute GVHD with no consistent survival advantage.
Dilanubicel is generated by ex vivo expansion of pooled donor CD34+ cells. The product demonstrated safety and support of early myelomonocytic recovery, leading to investigational use as a transient, third-party adjuvant graft, Milano and co-authors noted. Unlike other ex vivo expansion strategies that require real-time expansion of matched units, dilanubicel is an off-the-shelf, cryopreserved, T-cell-depleted product, derived from pooled cord blood donors.
For the current study, Milano and colleagues enrolled patients with acute leukemia in remission (n=25) or other hematologic malignancies. After myeloablative conditioning, each patient received a single HLA-matched cord blood unit, followed about 4 hours later by dilanubicel. All patients received GVHD prophylaxis with cyclosporine and mycophenolate mofetil.
The primary objective was to show that the addition of dilanubicel did not increase the risk of graft failure or relapse in the setting of single-unit cord blood transplantation. Secondary objectives included neutrophil and platelet engraftment, incidence of acute and chronic GVHD, and nonrelapse mortality.
The 28 patients had a median age of 36. Acute myeloid leukemia counted for 13 patients, acute lymphoblastic leukemia for 12, chronic myelogenous leukemia/myelodysplastic syndrome for two, and mixed-phenotype acute leukemia for one. All but six patients received a 4/6 HLA-matched cord blood unit and the rest received a 5/6 HLA-matched unit.
The results showed a 96% disease-free survival at last follow-up. As compared with 24 patients who underwent single- or double-unit cord blood transplants during the same timeframe, patients who received dilanubicel had faster neutrophil recovery (18 vs 22 days, P=0.03), earlier platelet engraftment (31 vs 37 days, P=0.01), and better overall survival (96% vs 66%, P=0.02).
In the control group, three patients died of infections, two of organ failure, and one of disease relapse. The incidence of grade 3/4 acute and 1-year chronic GVHD were 26% and 21%, respectively.
“We know that cord blood is a powerful stem cell source and for pretty much every patient,” said Milano. “We might now focus only on patients with high-risk disease to see if this approach can rescue these patients as well.”
Source link : https://www.medpagetoday.com/hematologyoncology/leukemia/121015
Author :
Publish date : 2026-04-29 14:10:00
Copyright for syndicated content belongs to the linked Source.