This article includes a first-person perspective from early breast cancer patient Lina. Lina was compensated for her time by Novartis.
Some patients with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative early breast cancer (eBC) may face a long-term risk of recurrence, even after surgery or initial treatment.1 What isn’t always part of the clinical conversation is how living with this long-term risk feels.
How Do Patients Feel About the Risk of Breast Cancer Recurrence?
Many breast cancer patients describe recurrence not as a loud fear, but as a quiet one that lingers long after treatment ends. That has certainly been true for me.
Breast cancer is rising fastest among women under 50.2 Working in health care, I understood recurrence risk intellectually. Still, nothing prepares you for the moment when statistics become personal. I was diagnosed with stage II HR+/HER2- eBC at age 40.
At the time, I felt healthy and active, balancing a full career and family life. Because of my age, a mammogram was not yet routine for me. But something didn’t feel right, so I kept advocating for one. That decision changed everything.
What followed was a whirlwind: chemotherapy, surgery, radiation. I focused on appointments and getting through each step. When treatment ended and everyone around me celebrated moving forward, a different reality set in: uncertainty.
Despite my knowledge, that uncertainty felt disorienting when it was my own life, my own family, and my own future.
According to the Novartis Breast Health & Experience Index, conducted by Harris Poll on behalf of Novartis, nearly 8 in 10 US women with breast cancer worry about recurrence, even after surgery and initial treatment end.3 Yet many of us hesitate to bring that fear into the exam room. I know I did.
During follow-up visits, I wanted to be seen as doing well and focused on moving forward. Talking about recurrence felt like reopening something I was supposed to have closed. But those fears don’t disappear simply because we stay quiet.
According to this same survey, nearly two-thirds of US women with breast cancer feel there is more they could be doing to help reduce their risk, and just as many wish they knew more about their treatment options.3
As I began looking ahead, conversations about my recurrence risk became central to my care. I learned that for some with HR+/HER2- eBC, recurrence risk can persist after surgery or initial treatment. Some experience recurrence within the first few years, while others remain at risk much longer. I was surprised to learn that having few or even no lymph nodes affected doesn’t always mean the risk is low.1,4
Risk of Breast Cancer Recurrence in HR+/HER2- eBC
Clinical studies show that up to 1 in 9 patients with node-negative disease (N0), and up to 1 in 4 patients with node-positive disease (4+ nodes or N2/N3) may recur within the first three years, and as many as half may recur within twenty years.1,4-6
KISQALI® (ribociclib) is indicated in combination with an aromatase inhibitor (AI) for the adjuvant treatment of adults with HR+/HER2- stage II and III eBC at high risk of recurrence.
Please see Important Safety Information below, and full Prescribing Information.
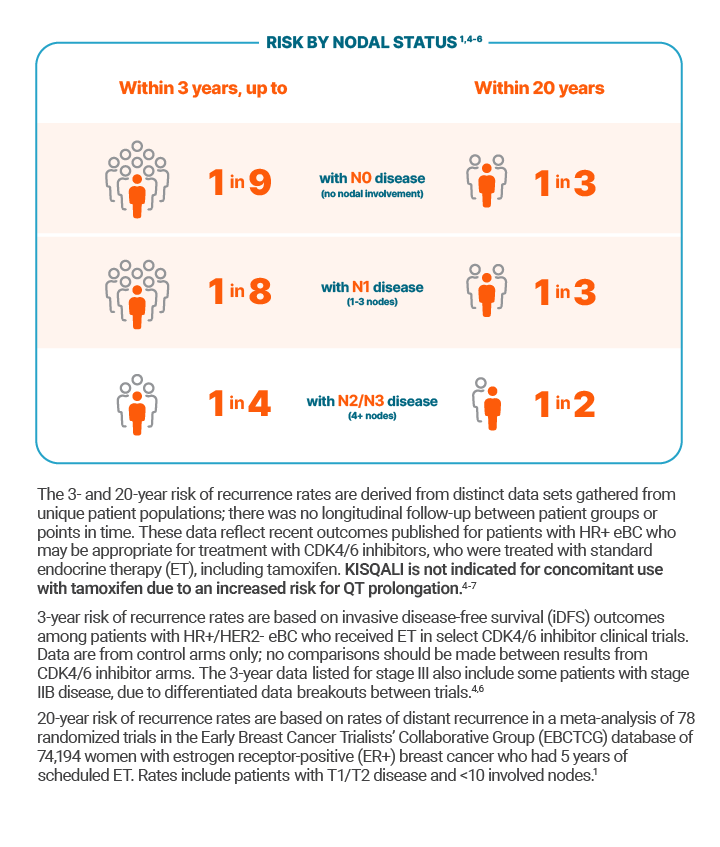
Given the long-term risk across disease stages, understanding recurrence patterns is critical when considering treatment strategies.
For people like me with high-risk eBC — meaning the cancer has features linked to a higher chance of recurrence, like spreading to nearby lymph nodes or, even without nodal spread, having more aggressive tumor features including certain genomic biomarkers — treatment options like KISQALI, a CDK4/6 inhibitor, added to a type of hormone therapy called an AI can be part of that discussion.
How Talking to My Doctor Helped Me Face My Fear
My oncologist was honest in explaining that managing risk now is different from treating metastatic disease later. Understanding that distinction helped me understand the role adjuvant therapy plays in eBC.
Starting treatment involved some trial and error, but the once-daily rhythm of KISQALI and built-in week off between cycles worked for me and helped me stay consistent. Just as important were the ongoing conversations with my oncologist. That partnership made treatment feel manageable.
Breast cancer changes more than your health. It changes how you think about time, risk, and the future. Many patients want to be active participants in their care but often rely on doctors to create space for those conversations.
Two years after my diagnosis, I am living a new normal. I still miss parts of the person I was before cancer, but I also recognize the strength that came from navigating uncertainty. The fear of recurrence does not disappear entirely, but understanding my options and knowing I am doing my part makes me feel empowered about my treatment plan.
If you are a doctor reading this, here is what I hope you know: many patients are thinking about recurrence long before they say it out loud. Creating space for those conversations can make an enormous difference. Sometimes what patients need most is permission to talk about the fears they are trying hard to carry quietly.
To learn more about recurrence risk in eBC and which patients may benefit from KISQALI, visit https://www.kisqali-hcp.com/early-breast-cancer/risk-of-recurrence.
Lina is currently being treated with KISQALI plus an AI for HR+/HER2- eBC and is a Voices of MORETM ambassador.
Novartis Pharmaceuticals Corporation has no control over third-party websites linked in this story, and makes no representation as to the accuracy, completeness, adequacy, or any other aspects of the information contained on such websites.
INDICATIONS
KISQALI is indicated:
- in combination with an aromatase inhibitor for the adjuvant treatment of adults with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative stage II and III early breast cancer (eBC) at high risk of recurrence
- for the treatment of adults with HR-positive, HER2-negative advanced or metastatic breast cancer (mBC) in combination with:
- an aromatase inhibitor as initial endocrine-based therapy; or
- fulvestrant as initial endocrine-based therapy or following disease progression on endocrine therapy
IMPORTANT SAFETY INFORMATION
Interstitial lung disease/pneumonitis. Severe, life-threatening, or fatal interstitial lung disease (ILD) and/or pneumonitis can occur in patients treated with KISQALI and other CDK4/6 inhibitors.
In patients with eBC (NATALEE) who received 400 mg KISQALI plus a nonsteroidal aromatase inhibitor (NSAI), 1.5% of patients had ILD/pneumonitis (grade 1/2).
In patients with advanced or mBC (MONALEESA-2, MONALEESA-3, MONALEESA-7), 1.6% of patients had ILD/pneumonitis of any grade, 0.4% had grade 3/4, and 0.1% had a fatal outcome. Additional cases of ILD/pneumonitis have occurred in the postmarketing setting, some resulting in death.
Monitor patients for pulmonary symptoms indicative of ILD/pneumonitis, which may include hypoxia, cough, and dyspnea. In patients who have new or worsening respiratory symptoms suspected to be due to ILD or pneumonitis, interrupt KISQALI immediately and evaluate the patient. Permanently discontinue treatment with KISQALI in patients with severe ILD/pneumonitis or any recurrent symptomatic ILD/pneumonitis.
Severe cutaneous adverse reactions. Severe cutaneous adverse reactions (SCARs), including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug-induced hypersensitivity syndrome (DiHS)/drug reaction with eosinophilia and systemic symptoms (DRESS) can occur in patients treated with KISQALI.
If signs or symptoms of SCARs occur, interrupt KISQALI until the etiology of the reaction has been determined. Early consultation with a dermatologist is recommended to ensure greater diagnostic accuracy and appropriate management.
If SJS, TEN, or DiHS/DRESS is confirmed, permanently discontinue KISQALI. Do not reintroduce KISQALI in patients who have experienced SCARs or other life-threatening cutaneous reactions during KISQALI treatment.
QT interval prolongation. KISQALI has been shown to prolong the QT interval in a concentration-dependent manner.
Avoid KISQALI in patients who are at significant risk of developing torsades de pointes (TdP), including those with:
- congenital long QT syndrome;
- uncontrolled or significant cardiac disease, recent myocardial infarction, heart failure, unstable angina, bradyarrhythmias, uncontrolled hypertension, high degree atrioventricular block, severe aortic stenosis, or uncontrolled hypothyroidism;
- electrolyte abnormalities;
- taking drugs known to prolong QT interval and/or strong CYP3A inhibitors as this may lead to prolongation of the QTcF interval.
Based on the observed QT prolongation during treatment, KISQALI may require dose interruption, reduction, or discontinuation.
In patients with eBC (NATALEE) who received 400 mg KISQALI plus NSAI, 8 out of 2494 patients (0.3%) had > 500 ms post-baseline QTcF interval value and 50 out of 2494 patients (2%) had > 60 ms QTcF increase from baseline. QTcF prolongation was reversible with dose interruption. The majority of QTcF prolongation occurred within the first 4 weeks of KISQALI. There were no reported cases of torsades de pointes.
In patients with advanced or mBC (MONALEESA-2, MONALEESA-3, and MONALEESA-7) who received 600 mg KISQALI plus NSAI or fulvestrant, 15 of 1054 patients (1.4%) had >500 ms postbaseline QTcF value, and 61 of 1054 (6%) had a >60 ms QTcF increase from baseline. QTcF prolongation was reversible with dose interruption. The majority of QTcF prolongation occurred within the first 4 weeks of KISQALI. There were no reported cases of torsades de pointes. In MONALEESA-2, in the KISQALI + letrozole treatment arm, there was 1 (0.3%) sudden death in a patient with grade 3 hypokalemia and grade 2 QT prolongation. No cases of sudden death were reported in MONALEESA-7 or MONALEESA-3.
Perform electrocardiogram (ECG) in all patients prior to starting KISQALI. Initiate treatment with KISQALI only in patients with QTcF values <450 ms. Repeat ECG at approximately Day 14 of the first cycle, and as clinically indicated.
Monitor serum electrolytes (including potassium, calcium, phosphorus and magnesium) prior to the initiation of KISQALI, at the beginning of the first 6 cycles, and as clinically indicated. Correct any abnormality before starting KISQALI.
Increased QT prolongation with concomitant use of tamoxifen. KISQALI is not indicated for concomitant use with tamoxifen. Avoid use of tamoxifen with KISQALI. In MONALEESA-7, the observed mean QTcF increase from baseline was >10 ms higher in the tamoxifen + placebo subgroup compared with the nonsteroidal aromatase inhibitor (NSAI) + placebo subgroup. In the placebo arm, an increase of >60 ms from baseline occurred in 6/90 (7%) of patients receiving tamoxifen, and in no patients receiving an NSAI. An increase of >60 ms from baseline in the QTcF interval was observed in 14/87 (16%) of patients in the KISQALI and tamoxifen combination and in 18/245 (7%) of patients receiving KISQALI plus an NSAI.
Hepatotoxicity. In patients with eBC and advanced or mBC, drug-induced liver injury and increases in transaminases occurred with KISQALI.
In patients with eBC (NATALEE) treated with KISQALI, drug-induced liver injury was reported in 9 patients (0.4%), of which 5 were grade ≥3 and 8 had resolved as of the data cutoff. There were 8 (0.3%) clinically confirmed Hy’s Law cases (including 4 out of 9 drug-induced liver injury mentioned above), 6 of which had resolved within 303 days and 2 were resolving, all after discontinuation of KISQALI. Grade 3/4 increases in alanine aminotransferase (ALT) and aspartate aminotransferase (AST) occurred in 8% and 4.7%, respectively, and grade 4 increases in ALT (1.5%) and AST (0.8%).
In patients with advanced or mBC (MONALEESA-2, MONALEESA-7, and MONALEESA-3) treated with KISQALI, grade 3 or 4 increases in ALT and AST occurred in 11% and 8%, respectively. Among the patients who had grade ≥3 ALT/AST elevation, the median time to onset was 92 days for the KISQALI plus aromatase inhibitor or fulvestrant treatment arms. The median time to resolution to grade ≤2 was 21 days in the KISQALI plus aromatase inhibitor or fulvestrant treatment arms. In MONALEESA-2 and MONALEESA-3, concurrent elevations in ALT or AST >3x ULN and total bilirubin >2x ULN, with normal alkaline phosphatase, in the absence of cholestasis (Hy’s Law) occurred in 6 (1%) patients and all patients recovered after discontinuation of KISQALI.
Perform liver function tests (LFTs) before initiating KISQALI. Monitor LFTs every 2 weeks for the first 2 cycles, at the beginning of each of the subsequent 4 cycles, and as clinically indicated. Based on the severity of the transaminase elevations, KISQALI may require dose interruption, reduction, or discontinuation.
Neutropenia. KISQALI causes concentration-dependent neutropenia. In patients with eBC (NATALEE) who received KISQALI plus NSAI, 94%, including 45% of grade 3/4, had a decrease in neutrophil counts (based on laboratory findings), 63% had an adverse drug reaction of neutropenia, and 0.3% had febrile neutropenia. The median time to grade ≥2 neutropenia was 18 days. The median time to resolution of grade ≥3 neutropenia to grade <3 was 10 days. Treatment discontinuation due to neutropenia was required in 1.1% of patients.
In patients with advanced or metastatic breast cancer (MONALEESA-2, MONALEESA-7, and MONALEESA-3) who received KISQALI plus NSAI or fulvestrant, 75% had neutropenia, 62% had grade 3/4 decrease in neutrophil count (based on laboratory findings), and 1.7% had febrile neutropenia. The median time to grade ≥2 neutropenia was 17 days. The median time to resolution of grade ≥3 neutropenia to grade <3 was 12 days. Treatment discontinuation due to neutropenia was required in 1% of patients.
Perform complete blood count (CBC) before initiating therapy with KISQALI. Monitor CBC every 2 weeks for the first 2 cycles, at the beginning of each of the subsequent 4 cycles, and as clinically indicated. Based on the severity of the neutropenia, KISQALI may require dose interruption, reduction, or discontinuation.
Embryo-fetal toxicity. Based on findings from animal studies and the mechanism of action, KISQALI can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise women of reproductive potential to use effective contraception during therapy with KISQALI and for at least 3 weeks after the last dose.
Adverse reactions in early breast cancer patients. Most common (incidence ≥20%) adverse reactions include infections, nausea, headache, and fatigue.
Laboratory abnormalities. In a clinical trial of patients with early breast cancer, the most common laboratory abnormalities reported in the KISQALI arm (all grades, pooled incidence ≥20%) were lymphocytes decreased, leukocyte decreased, neutrophil decreased, hemoglobin decreased, alanine aminotransferase increased, aspartate aminotransferase increased, creatinine increased, and platelets decreased.
Adverse reactions in advanced or metastatic breast cancer patients. Most common (incidence ≥20%) adverse reactions include infections, nausea, fatigue, diarrhea, vomiting, headache, constipation, alopecia, cough, rash, and back pain.
Laboratory abnormalities. Across clinical trials of patients with advanced or metastatic breast cancer, the most common laboratory abnormalities reported in the KISQALI arm (all grades, pooled incidence ≥20%) were leukocytes decreased, neutrophils decreased, hemoglobin decreased, lymphocytes decreased, AST increased, gamma-glutamyl transferase increased, ALT increased, creatinine increased, platelets decreased, and glucose serum decreased.
Please see full Prescribing Information.
References:
1. Pan H, Gray R, Braybrooke J, et al; EBCTCG. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med. 2017;377(19):1836-1846. doi:10.1056/NEJMoa1701830
2. American Cancer Society. Cancer Facts & Figures 2026. Atlanta, GA: American Cancer Society; 2026.
3. Harris Poll. Sponsored by Novartis Oncology. The Novartis Breast Health & Experience Index. 2025.
4. Mayer EL, Dueck AC, Martin M, et al. Palbociclib with adjuvant endocrine therapy in early breast cancer (PALLAS): interim analysis of a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2021;22(2):212-222. doi:10.1016/S1470-2045(20)30642-2
5. Pan H, Gray R, Braybrooke J, et al; EBCTCG. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med. 2017;377(19):1836-1846;(suppl). doi:10.1056/NEJMoa1701830
6. Johnston SRD, Toi M, O’Shaughnessy J, et al. Abemaciclib plus endocrine therapy for hormone receptor-positive, HER2-negative, node-positive, high-risk early breast cancer (monarchE): results from a preplanned interim analysis of a randomised, open-label, phase 3 trial. Lancet Oncol. 2023;24(1):77-90. doi:10.1016/S1470-2045(22)00694-5
7. Kisqali. Prescribing Info. Novartis Pharmaceuticals Corp.
4/26 FA-11596868
The MedPage Today Editorial team was not involved in the creation of this content.
Source link : https://www.medpagetoday.com/ad-insights/industry-clinic/120998
Author :
Publish date : 2026-05-11 15:00:00
Copyright for syndicated content belongs to the linked Source.