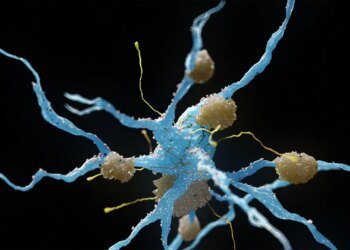
SAN DIEGO — Injecting the PD-1 inhibitor nivolumab (Opdivo) directly into high-risk oral precancers proved safe and reduced lesion size in the vast majority of patients enrolled in a small phase I study, allowing some to avoid surgery.
Among 29 patients with a high-risk premalignant oral lesion, 85% experienced a clinical response, with roughly two-thirds having their lesion size reduced by at least half over a median follow-up of 14.5 months from the first injection, Moran Amit, MD, PhD, of the University of Texas MD Anderson Cancer Center in Houston, reported here.
The cancer-free survival rate at 1 year was 76% among the complete responders, and no dose-limiting toxicities occurred, according to findings presented during a press briefing at the American Association for Cancer Research annual meeting.
Approximately 5% of the general population have precancerous oral lesions, which depending on the extent of dysplasia and other factors can carry up to a 36% risk of progressing to cancer, Amit explained.
Standard of care is surgery, but most patients experience recurrences requiring multiple procedures that can be debilitating — affecting speech, swallowing, articulation, and even feeding.
“That’s what we’re trying to avoid,” Amit said. “For the first time, we have a non-surgical, effective, and safe approach.”
All but four of the study participants completed all their treatment cycles and monitoring, despite a very heavy travel burden — one patient traveled 1,240 miles for treatment and the average one-way distance was 205 miles.
“That tells you how desperate they are — they don’t want [oral surgery] anymore,” Amit said. A placebo-controlled phase II trial is now underway at his institution. If the intralesional immunotherapy approach is validated, it could lead to treatment de-escalation, reduce surgical burden, and preserve quality of life, the researcher said.
The phase I trial enrolled 29 patients with at least one histologically confirmed, untreated premalignant oral lesion that had a high risk of progression to oral cancer due to its size, location, morphology, or extent of dysplasia, or the patient’s age or medical history.
More than half of the lesions were located on the tongue. Fifteen patients had lesions with high-grade dysplasia, while the remaining had low-grade (mild) dysplasia.
Patients were randomized to receive either 10 mg or 20 mg of nivolumab (representing 2% to 4% of a full 480-mg dose) injected directly into one of their oral lesions every 3 weeks, for a total of four cycles. Only one lesion per patient was treated in order to assess whether intralesional nivolumab’s effects would be systemic or restricted to the site of injection.
In terms of response, the median reduction in lesion area was 60%, and responses were observed for both low- and high-grade lesions. Twelve patients (41%) experienced histologic downgrading of their treated lesion, and 21% had a complete pathologic response.
At 1 year after starting treatment, 82% of treated lesions remained cancer free, and none of the patients with lesions that did not progress required surgery of the treated lesions. Six patients’ lesions progressed to cancer, but these were detected early and these lesions were resected.
Regarding safety, the most common adverse events were fatigue, diarrhea, and rash — well-known toxicities from immunotherapies such as nivolumab. There was one case each of grade 3 diarrhea, grade 3 hyperglycemia, and grade 4 acidosis. Mild injection-site reactions occurred in 40% of injections but resolved without intervention within 2 days.
Patient-reported outcomes demonstrated an improvement on almost every dimension, said Amit, including measures of “pain, articulation, swallowing, salivating, or other elements related to immunotherapy or head and neck cancer.”
Source link : https://www.medpagetoday.com/meetingcoverage/aacr/120908
Author :
Publish date : 2026-04-22 19:10:00
Copyright for syndicated content belongs to the linked Source.