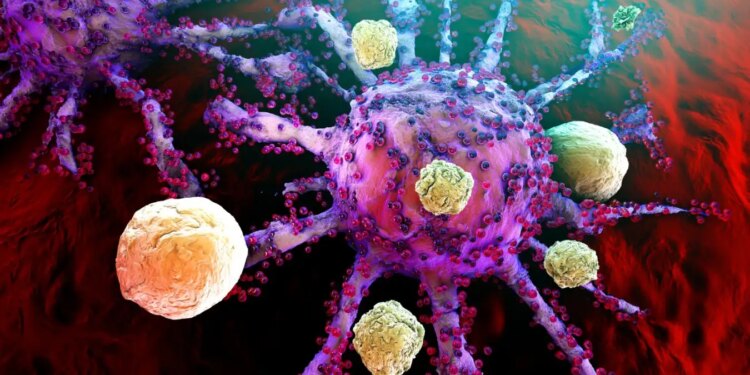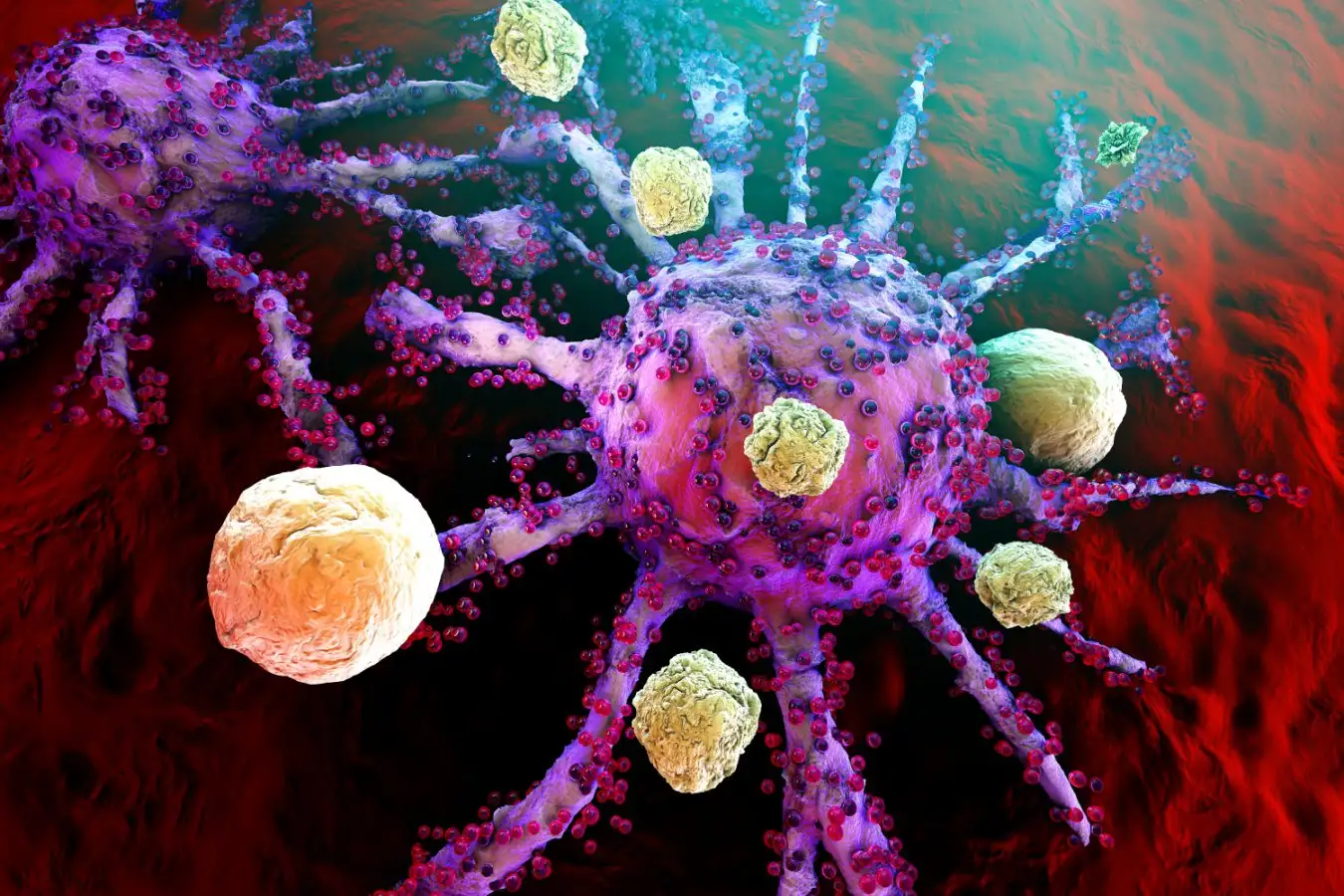
Illustration of T-cells attacking a growing cancer cell
Location South/Alamy
Our immune systems never stop targeting cells they regard as a threat, so it’s really bad news when rogue immune cells mistakenly turn on us, as they do in autoimmune conditions. Existing treatments suppress these attacks, but don’t stop them. But a new approach that addresses the cause of these disorders by killing off the rogue cells is proving wildly successful.
“All the big pharma companies are jumping on the bandwagon now,” says Reuben Benjamin at King’s College London. There are dozens of clinical trials under way around the world, and the first treatments could be approved as early as next year, he says, as they’re proving to be vastly superior to those currently used.
The key to these new treatments are genetically engineered cells known as CAR T-cells. These are made from the T-cells that your immune system usually employs to kill off invasive bacteria or virus-infected cells. The T-cells are extracted from a person, programmed to attack a specific kind of cell and then returned to that individual.
CAR T-cells were originally developed for treating cancers. They can do wonders, curing people when all other treatments have failed. But there are many issues with them. In particular, they only work for blood cancers, such as leukaemia, not for solid tumours. And they can trigger very serious side effects, such as brain inflammation, affecting speech and movement.
Cancers are caused by mutant cells that keep growing and dividing when they should stop. Autoimmune conditions are similar in that they are also caused by rogue cells, in this case ones that mistakenly attack a certain organ or cell type.
For instance, type 1 diabetes is caused by an immune attack on the cells in the pancreas that produce insulin. Multiple sclerosis is caused by an attack on the myelin sheaths that surround nerves. “The list of autoimmune conditions is huge,” says Benjamin.
After the body generates new immune cells in response to an infection, they undergo a screening process that should weed out any that mistakenly attack our bodies. But sometimes this process fails, and these rogue cells can then persist for the rest of our lives.
Why does this happen? A study out earlier this month has confirmed an idea first proposed in the 1950s, that these rogue cells have mutations in key genes involved in this screening process. The result is that they don’t kill themselves off when they should – meaning autoimmune conditions are even more similar to cancers than we thought.
Despite the similarity, it wasn’t obvious that the CAR T-cell treatments that work on cancers would work well for autoimmune conditions. One major issue is that rogue immune cells don’t stand out from the crowd, so, if you want to get rid of cells producing self-targeting antibodies, you have to kill off most antibody-producing cells, not just the rogue ones.
In people treated for cancers, CAR T-cells can persist for many years, which is useful in this case. But it seemed that using them to treat autoimmune conditions would leave a huge hole in people’s immune defences.
‘Miraculous’ results
Even with the risks, around five years ago, Fabian Müller‘s team at the University Hospital of Erlangen in Germany decided to go ahead and treat people with lupus using CAR T-cells.
“The first few patients were so sick that they would have died without the treatment,” says Müller. To the team’s astonishment, the CAR T-cells did their job and then disappeared after a few months, allowing the immune system to recover fully.
“We just were lucky, honestly,” says Müller. He thinks CAR T-cells persist in people treated for cancer because their immune systems are weakened: blood cancers are caused by immune cells growing out of control, so treatments target them. In people with autoimmune conditions, by contrast, the immune system is intact and recognises the CAR T-cells as foreign, killing them off.
Then there are those potentially deadly side effects – they’re just not happening in people treated for autoimmune conditions. “That’s really miraculous,” says Müller. For one thing, it changes the risk-vs-benefit analysis, meaning the approach won’t necessarily be limited to the most serious cases.
He thinks there are three explanations for the surprising findings. Firstly, CAR T-cells are killing huge numbers of cells in people with advanced cancers, but have far fewer cells to target in those with autoimmune conditions. Secondly, the quality of CAR T-cells from people with autoimmune disorders is higher, probably because they haven’t had treatments targeting cancerous immune cells. Finally, the immune systems of people with cancer may be releasing lots of alarm signals, causing the CAR T-cells to overreact.
Worldwide, it’s likely that several hundred people have now been treated for autoimmune diseases using CAR T-cells, and while trial results aren’t yet in, case reports suggest it is remarkably effective for treating conditions such as lupus, myasthenia gravis and ulcerative colitis. Doctors are cautious about calling it a cure, but where the CAR T-cells kill off all the rogue cells, these treatments very well could be that.
“In the cancer world, we tend to wait five years before we start talking about cures. In the autoimmune world, we just don’t know,” says Benjamin. Even if rogue cells do sometimes make a reappearance, people can be treated again.
Some caution is required. The damage wrought by self-targeting immune cells isn’t always reversible, even if their attacks stop. And not all rogue immune cells are easy to target, so it remains to be seen in how many autoimmune conditions this approach will work. That said, the list of conditions for which treatments are being developed even includes asthma, which isn’t really an autoimmune disorder.
The high cost of CAR T-cell treatments will be another factor limiting a wide rollout. Having to take cells from each individual, modify them and replace them is extremely expensive.
But there is reason for optimism. There are so-called off-the-shelf CAR T-cell treatments, where T-cells from a single donor are used to treat dozens of different patients. Off-the-shelf CAR T-cells didn’t work well in people with cancer, says Benjamin, because they didn’t survive that long in people’s bodies. But for autoimmune conditions, this could be an advantage.
Then there are “in vivo CAR T-cells”. This is where you turn T-cells into CAR T-cells inside a person’s body, rather than in a lab. This means everyone can get the same treatment, greatly reducing costs. “There’s massive excitement about this,” says Benjamin.
There could well be unexpected setbacks ahead, but, so far, using CAR T-cells for treating autoimmune conditions is working better than anyone dreamed five years ago. That’s fantastic news for the around 1 in 10 of us affected by them.
Topics:
Source link : https://www.newscientist.com/article/2524382-we-may-finally-have-a-cure-for-many-different-autoimmune-conditions/?utm_campaign=RSS%7CNSNS&utm_source=NSNS&utm_medium=RSS&utm_content=home
Author :
Publish date : 2026-04-28 09:00:00
Copyright for syndicated content belongs to the linked Source.